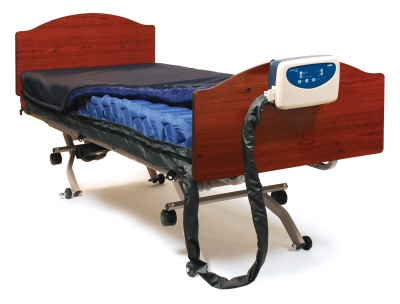
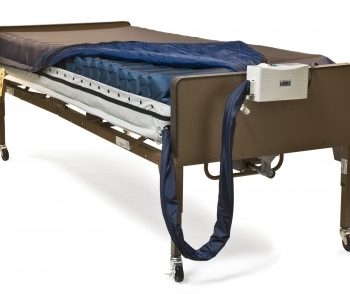
Simmons Clinical Sensor Plus
Alternating Pressure Mattress System with Active Sensor Technology
Intelligent, alternating pressure relief support surface that uses a proprietary Auto-Adjusting technology, used to automatically configure air pressure based on patient weight, relieving caregivers of any manual setup and assuring maximum comfort for the patient
Group II mattress system suitable for use in the prevention and treatment of pressure ulcers
Touch pad adjustment with LED indicators
Cycle Time: 10 minutes
Fine tuning function to manually adjust the air pressure settings
Four therapy settings including, Alternating pressure, Static, Alternating Seat Inflation or Static Seat Inflation
Lockable control panel to prevent unauthorized adjustment changes
Cell-on-cell design ensures that the mattress will hold air in the lower cells in the event of a power failure, preventing the user from bottoming out
Low pressure and power failure LED indicators and audible alarms
Quilted nylon PU cover is vapor permeable and fluid resistant
Quick CPR deflation is accomplished by simply disconnecting the quick-connect hose couplings from the control unit and/or releasing CPU tabs
Electrical rating: 120 VAC/ 60Hz
UL60601-1 and CSA C22.2 listed
Air input > 8 lpm
400 lb maximum weight capacity – EVENLY DISTRIBUTED
Designed to meet the requirements of HCPCS code: E0277
Meets CA Technical Bulletin #117 flammability requirements
1 year limited warranty
Not made with natural rubber latex
SIMMONS Clinical Care products manufactured by GF Health Products, Inc. under license from the trademark owner, Dreamwell, Ltd., a subsidiary of Simmons Bedding Company, Atlanta, Georgia.SIMMONS is a trademark of Dreamwell, Ltd. a subsidiary of Simmons Bedding Company.
$1,500.00
Simmons Clinical Sensor Plus, APM
78.7" x 35.4" x 8"
| Weight | 30 lbs |
|---|
Be the first to review “Simmons Clinical Sensor Plus”
Other Products From This Seller
$610.00
- Availability: in stock
$590.00
- Availability: in stock
$610.00
- Availability: in stock
$590.00
- Availability: in stock
$600.00
- Availability: in stock
$580.00
- Availability: in stock
$600.00
- Availability: in stock
$580.00
- Availability: in stock
$590.00
- Availability: in stock







Reviews
There are no reviews yet.